L’Oréal has introduced two light enabled beauty technology concepts at the Consumer Electronics Show in Las Vegas: a near infrared powered flat iron under the Light Straight platform and a silicone based LED eye mask targeting visible signs of under eye ageing. Commercial launch timelines are indicated for 2027 under brands within the company’s portfolio.
Light Straight Flat Iron: Infrared Assisted Thermal Management
The Light Straight and Multi Styler platform builds upon infrared assisted drying technology previously integrated into L’Oréal Professionnel’s AirLight Pro hair dryer. The new device applies near infrared energy in combination with controlled plate heating to reduce reliance on high surface temperatures during styling.
Reported technical characteristics include:
- Glass based plates
- Maximum plate temperature capped at approximately 320°F
- Claimed lower heat exposure relative to conventional straighteners
- Capability to create straight, wavy, or curled finishes
From an engineering standpoint, near infrared supplementation may enhance internal moisture management while reducing peak thermal stress on the cuticle. For device manufacturers, this signals continued convergence between cosmetic formulation science and electronics driven performance modulation.
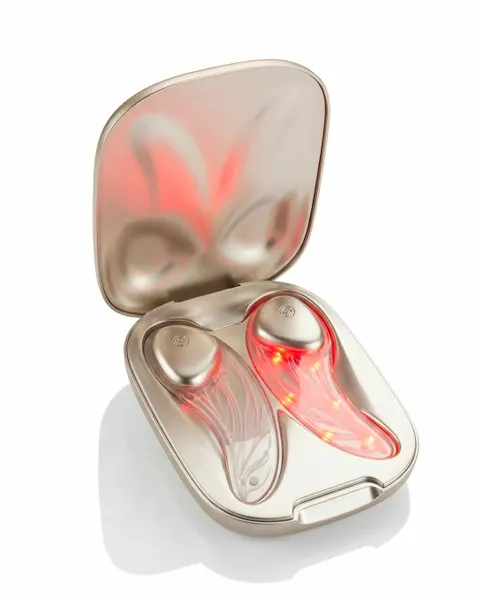
LED Eye Mask: Targeted Photobiomodulation Platform
The second innovation is a flexible LED eye mask constructed on a silicone substrate. The device integrates:
- Red light and near infrared light emission
- 16 LED chips per eye mask section, totaling 32 LEDs
- Time controlled 10 minute treatment cycles
Company disclosures indicate development of a full face configuration incorporating 208 LED chips designed to conform closely to facial contours.
Photobiomodulation devices using red and near infrared wavelengths are increasingly explored for collagen stimulation and microcirculation enhancement. However, regulatory classification may vary by jurisdiction depending on therapeutic versus cosmetic positioning.
Regulatory and Commercial Considerations
Devices integrating light based technologies may fall under different regulatory frameworks across markets:
- Cosmetic device classification if limited to aesthetic claims
- Medical device categorization if therapeutic or physiological claims are substantiated
- Electrical and consumer electronics compliance under safety standards
As launch approaches, positioning, claim substantiation, and risk assessment will determine market authorization pathways in the United States, European Union, and Asia Pacific jurisdictions.
Strategic Implications
L’Oréal’s introduction of these prototypes highlights:
- Increased integration of hardware and beauty science
- Expansion of at home device categories
- Continued R&D investment in augmented beauty technologies
- Cross disciplinary innovation between materials science, optics, and personal care
While still in the prototype stage, the announcements reinforce the growing relevance of energy based technologies within the premium beauty and personal care ecosystem.

